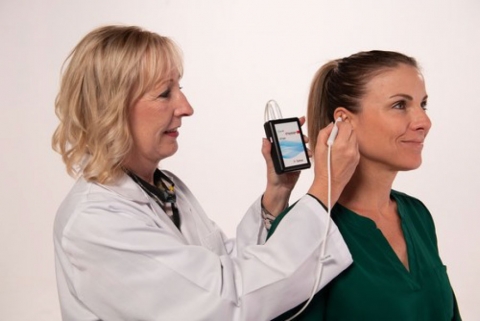
PALO ALTO, Calif. , July 28, 2022 /PRNewswire/ -- DyAnsys, Inc., has announced that PrimaryRelief, a percutaneous electrical neurostimulation device, has been clearedto treat painfollowing a Caesarean-section (C-section).
The percutaneous electrical nerve stimulator (PENS) system can be used for up to three days forsymptomatic relief of post-operative pain following a C-section delivery.
Primary Relief, a PENS device, is a signifi cant alternative to opioids forthe treatment of pain following a C-section.
"This device has been shown to make a difference for patients, effectively relieving pain withoutreliance on opioids or other analgesics. This is a signifi cant advancement in providing optionsto women," said DyAnsys Chief Executive Srini Nageshwar . "We look forward to connecting withphysicians and hospitals to providing this alternative to their patients."
The wearable, battery-operated device is designed to administer periodic low level electricalpulses to the ear over 72 hours from the activation of the device. The electrical pulses aredelivered to branches of the cranial nerves on the ear through a wire assembly and stimulationneedles.
The effectiveness of the device was demonstrated in a single-center, double arm, randomized,controlled, parallel assignment prospective study involving 44 participants who underwent C-section delivery. The analysis showed that minimally invasive nerve stimulation interventionusing Primary Relief device reduced the pain score faster than the standard analgesics. Noadditional opioid analgesics were required. No complications or adverse events were observedin any of the participants during the study period.
The nonclinical testing of Primary Relief device included biocompatibility testing, electricalsafety (electromagnetic compatibility and safety), performance bench testing and softwareverifi cation and validation.
DyAnsys offers two companion PENS devices - First Relief, with FDA clearance for the treatmentof diabetic neuropathic pain, and Drug Relief, with FDA clearance as an aid to drug withdrawal.
DyAnsys Inc. is a global company headquartered in California with subsidiaries in Switzerland and India . DyAnsys provides advanced medical diagnostic and monitoringsystems to clinicians in individual practices and hospitals. DyAnsys, ANSiscope. First Relief,Primary Relief and Drug Relief are registered trademarks, of DyAnsys, Inc.
Website: www.dyansys.com
For more information, contact Srini Nageshwar, (888) 950-4321
SOURCE DyAnsys Inc.

